Abstract
The present study investigated the effect of aqueous and ethanol extracts of Dialiumguineense stem bark on lipid profile and CCl4- induced histological changes in liver of Wistar rats. Adult male Wistar rats (n = 25) weighing 160 – 180 g (mean weight = 170 ± 10 g) were randomly assigned to five groups (5 rats per group): normal control, CCl4 control, silymarin, aqueous extract and ethanol extract groups. With the exception of normal control, the rats were exposed to CCl4 (single oral dose of 1.0 mL/kg body weight, bwt). Silymarin group rats were administered standard hepatoprotective drug, silymarin, at a dose of 100 mg/kg bwt, while those in the two treatment groups received 1000 mg/kg bwt of aqueous or ethanol extract orally for 28 days. Lipid profile parameters were determined in plasma, while rat liver was subjected to histopathological examination. The results showed that the levels of total cholesterol (TC), triacylglycerol (TG), high-density lipoprotein cholesterol (HDL-C), very-low density lipoprotein cholesterol (VLDL-C), low-density lipoprotein cholesterol (LDL-C) as well as atherogenic index of plasma (AIP) were significantly lower in CCl4 control group than in normal control group, but they were increased by extract treatment (p < 0.05). However, there were no significant differences in atherogenic coefficient (AC) and cardiac risk ratio (CRR) among the groups (p > 0.05). Carbon tetrachloride (CCl4) markedly disrupted the structure of hepatocytes and induced steatosis (intra-hepatocyte fat in-growth and inflammation) which was predominantly microvesicular. However, treatment with aqueous and ethanol extracts of D. guineense stem bark showed marked regeneration of hepatocytes (unremarkable hepatic lobular architecture). The toxic hepatic injury induced by CCl4 was significantly blocked by the plant extracts.
Author Contributions
Academic Editor: Anu Puri, National Institutes of Health, USA
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2022 Abu O.D, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Lipids are one of the necessary components which control cellular functions and homeostasis.
The liver plays an essential role in lipid metabolism, several stages of lipid synthesis and transportation 1. Therefore, it is reasonable to expect an abnormal lipid profile in severe liver dysfunction 1. Chemicals constitute an important cause of liver injury. Carbon tetrachloride is the most commonly used hepatotoxic agent for the induction of liver injuries in experimental animals 2, 3. There is a prominent decline in plasma total cholesterol and triacylglycerol levels in patients with severe hepatitis and hepatic failure because of reduction in lipoprotein biosynthesis 4.
Today, a substantial number of drugs are developed from plants which are active against a number of diseases 5.
Dialiumguineense is a medicinal plant used in Traditional Medicine for the treatment of infections such as diarrhea, severe cough, bronchitis, wound, stomachaches, malaria, jaundice, ulcer and hemorrhoids 6, 7. Extracts of the plant are reported to be rich in important phytochemicals 8, 9, 10. At present not much is known about the potential of extracts of D. guineense stem bark to alter lipid profile and histology of rats liver exposed to CCl4. This study investigated the effect of aqueous and ethanol extracts of Dialiumguineense stem bark on lipid profile and CCl4- induced histological changes in liver of Wistar rats.
Materials and Methods
Chemicals and Reagents
All chemicals and reagents used in this study were of analytical grade and they were products of Sigma-Aldrich Ltd. (USA).
Collection of Plant Material
The stem barks of D. guineense were obtained from Auchi, Edo State, Nigeria and authenticated at the herbarium of the Department of Plant Biology and Biotechnology, University of Benin, Benin City, Nigeria (No. UBHD330).
Plant Preparation and Extraction
The stem bark was washed and shade-dried at room temperature for a period of two weeks and then pulverized. Aqueous and ethanol extracts of the stem bark were obtained using cold maceration method as described previously 11.
Experimental Rats
Adult male Wistar rats (n = 25) weighing 160 – 180 g (mean weight = 170 ± 10 g) were obtained from the Department of Anatomy, University of Benin, Benin City, Nigeria. The rats were housed in metal cages under standard laboratory conditions: temperature of 25 oC, 55 – 65 % humidity and 12-h light/12-h dark cycle. They were allowed free access to rat feed (pelletized growers mash) and clean drinking water. Prior to commencement of the study, the rats were acclimatized to the laboratory environment for one week. Standard experimental protocol was followed for this study.
Experimental Design
The rats were randomly assigned to five groups (5 rats per group): normal control, CCl4 control, silymarin, aqueous extract and ethanol extract groups. With the exception of normal control, the rats were exposed to CCl4 (single oral dose of 1.0 mL/kg bwt) 11. Silymarin group rats were administered standard hepatoprotective drug, silymarin, at a dose of 100 mg/kg bwt, while those in the two treatment groups received 1000 mg/kg bwt of aqueous or ethanol extract orally for 28 days.
Blood Sample Collection and Preparation
At the end of the treatment period, the rats were euthanized. Blood samples were collected from the anesthetized rats via cardiac puncture in heparinized sample bottles, and centrifuged at 2000 rpm for 10 min to obtain plasma which was used for biochemical analysis. The liver of all experimental rats were harvested, washed in ice – cold saline, blotted dry and placed in plain containers. Weighted portions of the liver were placed in 10 % phosphosaline (pH 7.0) for histological examination.
Biochemical Analysis
Histological Examination of the Tissues
Portions of the liver were serially sectioned and fixed in 10 % formalin for 48 h. The specimen was then dehydrated using varied concentrations of ethanol and cleared in three changes of xylene before embedment in paraffin. Serial sections (4 μm thick) were made and stained with haematoxylin and eosin (H & E) according to standard method. Histological assessment was performed under light microscopy. In every H and E section, a minimum of 25 circular tubules were measured in two axes drawn perpendicular to each other using an image analyzer (Image Proplus, version 3.0).
Statistical Analysis
Count data are expressed as mean ± SEM (n = 5). Statistical analysis was performed using SPSS (version 20). Groups were compared using Duncan multiple range test. Statistical significance was assumed at p < 0.05.
Results
Effect of Extracts of D. guineense Stem Bark on Relative Organ Weight
There were no significant differences in relative organ weight among the groups p > 0.05;Table 1.
Table 1. Relative Organ Weights of Rats| Group | Relative organ weight x 10-2 |
| Normal Control | 2.98 ± 0.05 |
| CCl4 Control | 2.86 ± 0.06 |
| Silymarin | 2.84 ± 0.06 |
| Aqueous Extract | 2.98 ± 0.05 |
| Ethanol Extract | 2.99 ± 0.20 |
Effect of Extracts of D. guineense Stem Bark on Lipid Profile of Rats
The levels of TC, TG, HDL-C, VLDL-C, LDL-C as well as AIP were significantly lower in CCl4 control group than in normal control group, but they were increased by extract treatment (p < 0.05). There were no significant differences in AC and CRR among the groups (p > 0.05). These results are shown in Table 2, Table 3, Table 4.
Histology of normal control rat liver revealed distinct centriole with the hepatocytes and well fenestrated sinusoidal with mild mononuclear cells, while that of CCl4 control showed visible centriole with the hepatocytes nuclei appearing vacuolated. There was mild fatty changes and visible mononuclear cells. Histopathological examination of silymarin group rats liver revealed congested centriole with fairly pyknotic nuclei hepatocytes and well fenestrated sinusoidal with mild mononuclear cells. Similarly, histological changes in aqueous extract-treated rats revealed congested centriole with thickened wall surrounded by mononuclear cells. The hepatocytes had pyknotic nuclei, while those of ethanol extract-treated rats showed congested centriole with thickened wall surrounded by mild mononuclear cells with the hepatocytes and well fenestrated sinusoidal.. Figure 1
Table 2. Comparison of Lipid Profile Parameters Among the Groups| Group | TC (mg/dL) | TG (mg/dL) | HDL-C (mg/dL) |
| Normal Control | 190.34 ± 29.43 | 65.27 ± 7.63 | 22.61 ± 2.17 |
| CCl4 Control | 137.36 ± 7.85 | 28.59 ± 3.82 | 15.60 ± 2.03 |
| Silymarin | 180.53 ± 17.99a | 46.69 ± 2.43a | 25.32 ± 4.07a |
| Aqueous Extract | 176.61 ± 12.62a | 47.17 ± 0.00a | 21.89 ± 0.00a |
| Ethanol Extract | 200.00 ± 5.99a | 66.70 ± 4.21a | 24.99 ± 2.16a |
| Group | VLDL-C (mg/dL) | LDL-C (mg/dL) | AIP |
| Normal Control | 13.06 ± 3.53 | 154.67 ± 7.63 | 155.25 ± 7.21 |
| CCl4 Control | 5.72 ± 0.76 | 116.04 ± 3.82 | 116.41 ± 4.13 |
| Silymarin | 9.34 ± 2.29a | 145.87 ± 2.43a | 146.24 ± 4.07a |
| Aqueous Extract | 9.43 ± 0.00a | 145.29± 0.00a | 145.72 ± 0.00a |
| Ethanol Extract | 13.34 ± 4.29a | 161.67 ± 4.21a | 162.20 ± 6.12a |
| Group | AC | CRR |
| Normal Control | 7.42 ± 0.53 | 8.42 ± 0.63 |
| CCl4 Control | 7.81 ± 0.76 | 8.81 ± 0.82 |
| Silymarin | 6.13 ± 0.92 | 7.13 ± 0.93 |
| Aqueous Extract | 7.07 ± 0.75 | 8.07 ± 0.94 |
| Ethanol Extract | 7.00 ± 0.79 | 8.00 ± 0.77 |
Figure 1.Effect of Extracts of D. guineense Stem Bark on Histology of Rats Liver.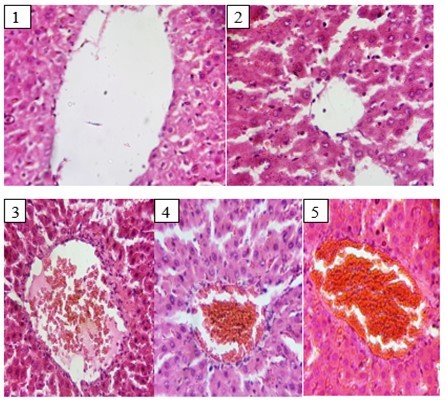
Discussion
The aim of this study was to investigate the effect of aqueous and ethanol extracts of D. guineense stem bark on lipid profile and CCl4- induced histological changes in liver of Wistar rats.
Lipid profile is a panel of blood tests that serves as an initial broad medical screening tool for the assessment of abnormalities in the concentrations of lipids, such as cholesterol and triacylglycerol. These tests can identify certain genetic diseases and determine approximate risks for cardiovascular diseases (CVDs), certain forms of pancreatitis, and other diseases. Lipid profile typically includes LDL-C, HDL-C, TG, TC, VLDL-C and CRR 12. Abnormal lipid profile is seen in those with severe liver dysfunction 4. There is a prominent decline in plasma TC and TG levels in patients with severe hepatitis and hepatic failure because of reduction in lipoprotein biosynthesis 4. The results obtained in this study suggest that the lipids synthesis ability of the liver may be reduced with CCl4 induction, and are in agreement with those of previous reports 18, 19, 20. It is likely aqueous and ethanol extracts of D. guineense stem bark regulated liver secretion and uptake of plasma lipoproteins. The ability of the extracts to promote lipids biosynthesis could be due to the enhanced transport of acetate into the liver cell, resulting in increased substrate (acetate) availability. Elevated levels of serum TC and TG in CCl4 treated rats has been reported 21, 22.
In this study, the effect produced by the extracts of the medicinal plant was comparable to that of silymarin (standard hepatoprotective drug). Silymarin protects liver against xenobiotic injury by controlling the liver secretion and uptake of plasma lipoprotein, while increasing the intracellular glutathione content 23. Silymarin plays the role of an anti-inflammatory agent, through its ability to inhibit neutrophil infiltration and regulate the release of inflammatory mediators. It has been reported that silymarin prevents CCl4-induced lipid peroxidation and hepatotoxicity in mice, first, by decreasing the metabolic activation of CCl4 and second, by acting as a chain-breaking antioxidant 24. In addition, silymarin is able to stimulate protein synthesis resulting in production of new liver cells to replace older and damaged ones 25.
Histopathological studies provided supportive evidence for lipid profile analysis. Administration of CCl4 showed marked disruption of the structure of hepatocytes, induced steatosis (intra-hepatocyte fat in-growth and inflammation) which was predominantly microvesicular. However, treatment with aqueous and ethanol extracts of D. guineense stem bark showed marked regeneration of hepatocytes (unremarkable hepatic lobular architecture), which slightly affected the normal architecture of hepatocyte cords with few areas of discontinuity. Similarly, treatment with silymarin induced mild portal congestion and dilatation without any evidence of steatosis.
Conclusion
The toxic hepatic injury induced by CCl4 was significantly blocked by treatment with aqueous and ethanol extracts of D. guineensestem bark. The extent of improvement in lipid profile of the rats may not be unconnected with the dose used. Further studies are needed to assess the predictive values of measuring lipid profile as a means to estimate the extent of liver damage in cirrhotic rats.
References
- 1.S E Friedman, J H Grendel, K R McQuaid. (2003) Current Diagnosis and Treatment in Gastroenterology. , New York 664-679.
- 2.A M Gupta, U K Mazumder, Das S. (1994) Effect of alkaloid extractsClerodendroncolebrookianumon Hematological parameters and hepatorenal function in mice.Indian. , J. Exp. Biol 32, 189-191.
- 3.R S Cotran, Kumar V, Fausto N, Nelso F, S L Robbins et al. (2005) Robbins and Cotran pathologic basis of disease St. , Louis, MO 878.
- 5.D S Fabricant, N R Farnsworth. (2001) The value of plants used in Traditional medicine for drug discovery.Environ HealthPerspect.109: 69. 75.
- 6.O D Abu, K E Imafidon, M E Iribhogbe. (2015) Biochemical effect of aqueous leaf extract ofIcacinatrichantaOliv. on urea, creatinine and kidney oxidative status in CCl4-induced Wistar rats.Nigerian. , Journal of Life Sciences 5(1), 85-89.
- 7.Bero J, Ganfon H, M C Jonville, Frederich M, Gbaguidi F et al.(2009).In vitroantiplasmodial activity of plants used in Benin in traditional medicine to treat malaria.Journal of Ethnopharmacology. 122(3), 439-444.
- 9.Kar A. (2007) Pharmacognosy and Pharmaco-biotechnology (Revised-Expanded Second Edition). , New Delhi 332-600.
- 10.O D Abu, I O Onoagbe, Obahiagbon O. (2020) Qualitative phytochemical screening and proximate analysis ofDialiumguineensestem bark.IAR. , Journal of Agriculture Research and Life Sciences 1(4), 108-112.
- 11.O D Abu, K E Imafidon, M E Iribhogbe. (2017) Aqueous leaf extract ofIcacinatrichantaOliv.ameliorates CCl4-induced liver toxicity. in Wistar rats.Journal of the NigerianSociety of Experimental Biology.17 3, 107-111.
- 12.Reiser R, J L Probstfield, Silvers A, L W Scott, M L Shorney et al. (1985) Plasma lipid and lipoprotein response of humans to beef fat, coconut oil and safflower oil. , Am J Clin Nutr 42(2), 190-197.
- 13.N W Tietz, P R Finley, E L Pruden. (1990) . Clinical Guide to Laboratory Tests. 2nd Edition, W.B , Philadelphia. Pp 304-306.
- 14.M F Lopes-Virella, Stone P, S andColwell Ellis, A J. (1977) Cholesterol determination in high-density lipoproteins separated by three different methods.Clin Chem. 23(5), 882-884.
- 15.W T Friedewald, Levy R I andFredrickson, S D. (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge.Clin Chem. 18(6), 499-502.
- 16.C J Ikewuchi, Ikewuchi C. (2009) . Alteration of Plasma Lipid Profiles and Atherogenic Indices by Stachytarpheta jamaicensis L.Vahl).Biokemistri 21(2), 71-77.
- 17.Frohlich J, Dobiasova M. (2003) . Fractional Esterification Rate of Cholesterol and Ratio of Triglycerides to HDL-Cholesterol Are Powerful Predictors of Positive Findings on Coronary Angiography.Clinical Chemistry.49 11, 1873-1880.
- 18.Siagris D, Christofidou M, G J Theocharis, Pagoni N, Papadimitriou C et al. (2006) Serum lipid pattern in chronic hepatitis C: histological and virological correlations. , J ViralHepat 13(1), 56-61.
- 19.M A Selimoglu, Aydogdu S, R V Yagci. (2002) Lipid parameters in childhood cirrhosis and chronic liver disease. , PediatrInt 44(4), 400-403.
- 20.A C Sposito, C G Vinagre, F L Pandullo, Mies S, Raia S et al. (1997) Apolipoprotein and lipid abnormalities in chronic liver failure. , BrazJ Med Biol Res 30(11), 1287-1290.
- 21.Venkatanarayana G, Sudhakara G, Rajeswaramma K, Indira P. (2013) Combined effect of curcumin and vitamin E against CCl4-induced liver injury in rats.American. , Journal of Life Sciences.1 3, 117-124.
- 22.Boll M, L W Weber, Becker E, Stampfl A. (2001) Hepatocyte damage induced by carbon tetrachloride: Inhibited lipoprotein secretion and changed lipoprotein composition.Naturforsch. 56(3), 283-290.
- 23.H Z Toklu, Akbay T, Velioglu-Ogunc A, F N Ercan, Keyer-Uysal M et al. (2008) Silymarin, the Gedik, antioxidant component ofSilybummarianum, prevents sepsis-induced acute lung and brain injury.Journal of Surgery Research. 145(2), 214-222.
- 24.Letteron P, Labbe G, Degott C, Berson A, Fromenty B et al. (1990) Mechanism for the protective effects of silymarin against carbon tetrachloride-induced lipid peroxidation and hepatotoxicity in mice: Evidence that silymarin acts as an inhibitor of metabolic activation and as a chain-breaking antioxidant.Biochemical Pharmacology.39.
Cited by (1)
- 1.Ogbuewu Ifeanyichukwu Princewill, Mbajiorgu Christian Anayo, 2023, Dietary Dialium guineense stem-bark supplementation improves growth performance and haemato-biochemical characteristics of broiler chickens, Heliyon, 9(6), e17341, 10.1016/j.heliyon.2023.e17341
