Abstract
Two samples of the defectdouble perovskites with general formula Sr2CaNbO5.5 and Sr2CaTaO5.5 were synthesized and their cap abilities in the removal of neutral red dye investigated. Both samples have faced cubic perovskite-type structure in space group Fm3m. The replacement of Nb5+ by Ta5+ has influenced both crystal size and density of the two oxides but showed no impact on the removal capacities. The crystallite size of Sr2CaNbO5.5 and Sr2CaTaO5.5 was determined to be 93.56 and 43.69 nm respectively. The maximum removal capacities of Neutral red are found to be 190.5 mg/g using both the two oxides. This result is consistent with the cell volumes which displayed similar values. The removal of Neutral Red has a negative relationship with temperature but steadily increases as pH increased.
Author Contributions
Academic Editor: Mohammad Tavakkoli Yaraki, National University of Singapore, Singapore.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Labib. A. Awin, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Organic dyes are considered to be extremely environmental pollutants. Their effluents are, in many cases, carcinogenic and toxic1. They consist of two main groups: chromophores which are responsible for colour and auxochromes that are responsible for intensity of the colour. Dyes are classified according to chemical structure and type of application. Based on the chromophore, 20–30 different groups of dyes can be discerned, with azo, anthraquinone, phthalocyanine and triarylmethane accounting for the most important groups2. The emissions of dyes from industrial activities causes degradation of water quality, thus many methods are reviewed to remove such organic pollutants. These methods can be divided to physical processes such as adsorption, chemical treatments such as chemical oxidation and other such as biological degradation technique3, 4. Adsorption has become more popular for wastewater treatment due to their efficiency in the removal. It can produce economically high quality water is sufficient time5, 6.
Recently, there is a growing interest in developing new adsorbent materials with diverse compositions, properties and functionalities. The physical characteristics of the adsorbents, such as surface area, porosity, size distribution, density and surface charge, all influence the adsorption process. Perovskite oxides with general formula ABO3 are promising candidates for dyes adsorption. They are highly versatile due to the flexibility in the chemical composition with a large number of cations7. Those cations can fit into both the A and B positions within the same crystalline structure. Perovskite oxides that contain a lanthanide element at position A and a transition metal at position B are used more frequently in heterogeneous catalysis, obviously exploiting the catalytic properties of transition metals. Perovskites with oxygen and cation non-stoichiometry can achieve the desired catalytic activity, including multifunctional catalytic properties7, 8. This work investigates the capability of the defect double perovskite Sr2CaNbO5.5 and Sr2CaTaO5.5 in the removal of Neutral Red dye from aqueous solutions. In such double perovskite structure, it is anticipated that the two smallest cations will order in the octahedral sites, this ordering being a consequence of the differences in the size and/or charge between the two cations. The largest cation will then occupy the 12-coordinate (cuboctahedral) site. The corresponding ionic radii of Sr2+ (12 coordinate ionic radius, 1.44 Å and 6 coordinate ionic radius. 1.18 Å) 9 Ca2+ (1.34 and 1.00 Å), Nb5+ (6 coordinate ionic radius. 0.64 Å)and Ta5+ (0.64 Å)cationssuggest that the (Nb5+, Ta5+) and one Ca2+ cation will occupy the 6-coordinate sites whereas the Sr2+ will occupy the cuboctahedral sites.
Nuteral Red is a eurhodin dye used for staining in histology. It is used as a counter stain in combination with other dyes, and for many staining methods. Together with Janus Green B, it is used to stain embryonal tissues and supravital staining of blood2.
Experimental
Sample Preparation
The preparation of samples involved different stoichiometric compositions of Ta2O5 (Merck, 99.99%) and CaCO3 or SrCO3 (BDH, 99.98-99.99%). The mixtures were initially ground and preheated at 850°C for 12 h, and then reground and heated at 1100°C for 48 h.
Instrumentations
The crystallography of the samples was examined by a PANalytical X’Pert X-ray powder diffraction using Cu Kα radiation (1.5400 Ȧ) and a PIXcel solid-state detector. the operating voltage was 40kV and the current was 30 mA. he samples were measured in lat plate mode at room temperature with a scan range of 10°<2θ<80° and a scan length of 10 mins were used. The structures were refined using the program RIETICA10.
The absorbance of solutions was determined using ultraviolet visible spectrophotometer (UV/Vis, model Spect-21D) and (190-900 Perkin- Elmer) at maximum wavelength of absorbance (520 nλ). The concentrations of solutions were estimated from the concentration dependence of absorbance fit. The pH measurements were carried out on a WTW720 pH meter model CT16 2AA (LTD Dover Kent, UK) and equipped with a combined glass electrode.
Batch Mode
Batch mode removal studies were carried out by varying several parameters such as contact time, pH, temperature and mass of prepared oxide (adsorbent). Essentially, a 50 ml of dye solution with concentration of 10 ppm was taken in a 250 ml conical flask in which the initial pH was adjusted using HCl/NaOH. Optimized amount of adsorbent was added to the solution and stirred using magnetic stirrer for specific time. The oxide samples were separated from solutions using centrifuge 3500 CPM for 5 minutes.
Result and Discussions
Characterization of Oxides
The preparation of an extensive solid state solution allows us to study the effects of altering the chemical composition on the physical properties. The heating regime described above produced crystalline, yellowish coloured, samples. X-ray diffraction measurements (Figure 1) show the samples to be highly crystalline and the data are consistent with these all having a faced cubic structure with space group (Fm3m). The solid state chemistry of Ta5+ and Nb5+ are generally believed to be similar, so it is reasonable to expect Sr2CaTaO5.5 will display similar behaviour to Sr2CaNbO5.5. The cell volume of Sr2CaNbO5.5 (559.49 Å3) is found to be similar to that for Sr2CaTaO5.5 (557.67 Å3). This reflects the similarity in the ionic radii of both Nb5+ (0.64 Å)and Ta5+ (0.64 Å)cations.
Figure 1.The XRD patterns of Sr2CaNbO5.5 and Sr2CaTaO5.5.
The Average Crystallite size Dp, specific surface area S, lattice strain φ, Lattice parameter a and Cell volume V estimated from X-ray diffraction data are summarised in Table 1. The crystallite size can be calculated using sheerer formula 11 (Equation. 1) where the specific surface area can be calculated using Sauter formula 12 (Equation.2) in which ρ is the density of the synthesised material.
Dp= (0.94λ)/(β1/2×cosϴ). (1)
S = 6000/ (Dp ×ρ). (2)
Both the shape and the size of particles are defined by the preparation methods; however the ratio of nucleation to growth rates of particles is also important. Each of these processes depends in turn on variations in the reaction conditions such as the temperature, the nature and concentrations of metal and ligand, and the nature of stabilizer and reducer. The differences in the crystallite size, surface area and density of the two oxides (see table) could be attributed to one of these variations.
Table 1. Average Crystallite size Dp, Specfic surface area S, lattice strain φ, Lattice parameter a and Cell volume V. estimated from X-ray diffraction data.| Formula | D p ( ηm ) | ρ(g/cm 3 ) | S (m 2 /g) | φ | a (Å) | V (Å3) |
| Sr2CaNbO5.5 | 50.45 | 4.70 | 13.64 | 0.0015 | 8.2401(2) | 559.490(1) |
| Sr2CaTaO5.5 | 43.69 | 5.67 | 24.20 | 0.0031 | 8.2300(3) | 557.670(1) |
Batch Mode
Effect of Time
The removal percentage of dyes over the adsorbents can be calculated as: R% = [(Ci-Ct)/Ci] × 100, where R% is the removal percentage, Ci = 10 ppm is initial concentration of dye solution, Ct is the concentration of dye at contact time estimated from the concentration dependence of absorbance fit. Figure 2. shows the time dependence of NR removal at room temperature. There is no finite time was observed for the dye removal up to 180 min. The removals of the dye increase as the contact time increases. The removal of NR on the surface of Sr2CaNbO5.5 and Sr2CaTaO5.5 were found to be similar (~94%). This result neglects the impact of the crystallite size on the adsorption properties of the two oxides. The removal of the organic ye is likely affected by the chemical compositions of the oxides. The chemistry of Nb+5 and Ta+5 cations is anticipated to be similar.
Figure 2.The time dependence of NR removal at room temperature. The volume, concentration and pH of the dyes solution are 50ml, 10ppm and 5.1 respectively.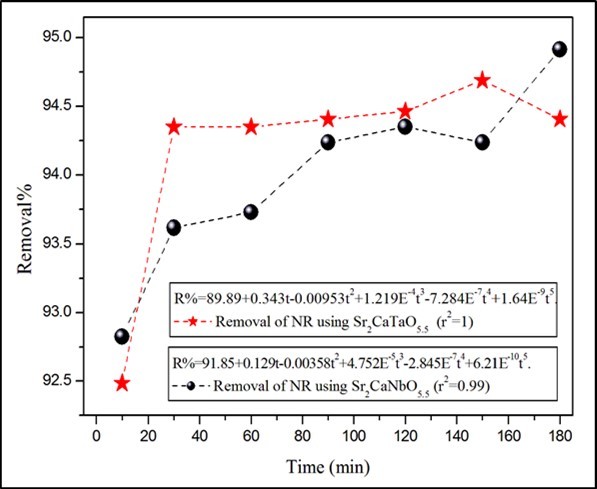
Figure 3 illustrates the wavelength dependence of absorbance for NR solutions at room temperature using Sr2CaNbO5.5 and Sr2CaTaO5.5. The maximum wavelength of NR was shifted from 520 nm to 440 nm suggesting organic degradation of the dye probably occurred.
Figure 3.The wavelength dependence of absorbance for NR solution. The time, volume, concentration, adsorbent mass and pH of NR solution are 180 min, 50ml, 10ppm, 0.1 g and 5.1 respectively.
Effect of Adsorbent Mass
The amount of the dye adsorbed by one gram of the oxides (Q) was calculated as following: Q (mg/g) = [(Ci-Ct)×V]/W, where t= 180 min is the contact time, V= 50 ml is the volume of NR solution and W is the mass of oxides. As shown in Figure 4, Q decreases as the mass of adsorbents increased. The maximum capacity of adsorbent Qmax can be estimated from the intercept of the liner fit of 1/Qt at Y axis. Both Sr2CaNbO5.5 (50.4 nm, 13.6 m2/g ) and Sr2CaTaO5.5 (43.7 nm, 24.2 m2/g) exhibited a similar value of Qmax(190.5 mg/g) at room temperature. This result is consistent with the similarity in the cell volumes of the two oxides which determined to be (~ 558 Å3). The removal capability of Neutral Red is higher than those recorded for Methyl violet when the perovskite oxides BaSr2NbO5.5 (~9.3 mg/g)13 and Sr2CaNbO5.5 (47.39 mg/g )14 were used as adsorbents.
Figure 4.The effect of adsorbent mass on the removal. The time, volume, concentration and pH of dye solutions are 150min, 50ml, 10ppm and 5.1 respectively.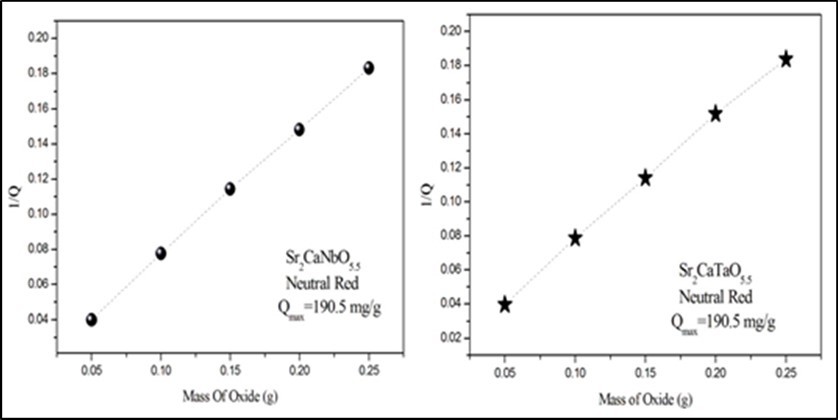
Effect of Temperature
Temperature has an important impact on the adsorption process. An increase in temperature helps the reaction to compete more efficiently with e–/H+ recombination. The removal of NR dye was investigated at 25, 40, 60 and 100oC. The obtained results are illustrated below in Figure 5. The removal of NR dye decreased as temperature increased. For instance, the removal of NR decreased from ~94% at 25ᴼC to ~74% at 100ᴼC. This result is disagreed with normal expectations, and is probably a consequence of an increase in the thermal energy. It is anticipated that higher temperature induces higher mobility of active sites of adsorbents and the surface area is decreased by increase the temperature15. The energy of activation (Ea), was calculated from the Arrhenius plot of ln R vs 1000/T. Arrhenius plot shows that the activation energies of the removal are negative and equal to 2.830 and 2.948 kJ/mole for Sr2CaTaO5.5 andSr2CaNbO5.5 respectively. This reflects the similarity in the strength of the interaction forces between the dye and the two oxides.
Figure 5.The effect of temperature on the MV removal. The time, volume, pH and concentration of dyes solutions are 150min, 50ml, 5.1 and 10ppm respectively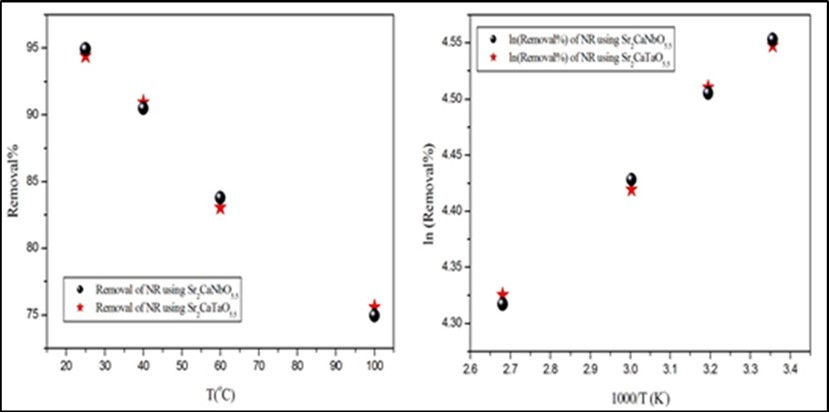
Effect of pH
The pH of solutions is a key parameter in dye adsorption. The magnitude of electrostatic charges which are impacted by the ionised dye molecules is controlled by the solution pH. As a result the rate of adsorption will vary with the pH of the medium used. In general, at low solution pH, the percentage of dye removal will decrease for cationic dye adsorption, while for anionic dyes the percentage of removal will increase. This is due to the increase in the positive charge on the solution interface and the adsorbent surface. In contrast, high solution pH is preferable for cationic dye adsorption but shows a lower efficiency for anionic dye adsorption. The positive charge at the solution interface will decrease while the adsorbent surface appears negatively charged 16.
To study the effect of pH, experiments were carried out at various pH values, ranging from 2 to 10 for constant dye concentration (10 ppm) and adsorbent mass (0.1g). Figure 6 presents the removal of dyes as a function of pH. It was observed that the removal of NR using the two oxides slightly increases as pH inceased. The highest removal of the dye was recorded at pH= 10 around 94 % where the lowest removal was recorded at pH= 2 around 93%. The removal efficiency of the two adsorbents seems to be similar in the acid media where in the basic media small differences (≤ 0.9%) can be noticed.
Figure 6.The effect of pH on the removal of NR. The time, volume and concentration of dyes solution are 180 min, 50ml and 10ppm respectively.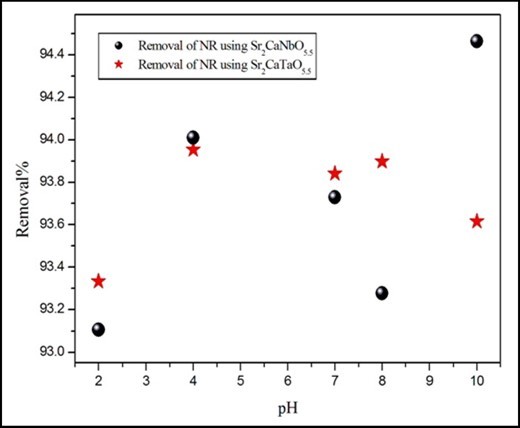
Conclusion
The removal of Neoutral Red from aqueous solution by the nano particle oxides Sr2CaNbO5.5 and Sr2CaTaO5.5 has been reported. The oxides were synthesised by solid state reaction and characterized by XRD. Despite the differences in crystallite size, the mounts of NR adsorbed by the two oxides were similar. The replacement of Nb5+ by Ta5+ has showed no influence on the removal capacities. The maximum removal capacities of Neutral red are 190.5 mg/g for the two oxides. Such result could be attributed to the similarity in the cell volumes of the oxides. The removal of dye gradually increased as time and pH increases but decreased as temperature increased.
References
- 3.Cheremisinoff N.. P.(2002) Handbook of Water and Wastewater Treatment Technologies, Butter worth-Heinemann .
- 4.Tushar Kanti Sen (2015) Physical, chemical and biological Treatment Process for Water and Wastewater. , Nova
- 7.Junjiang Zho etal (2014) Perovskite Oxides: Preparation, Characterizations and Applications in Heterogeneous Catalysis. , ACS Catal 4(9), 2917-2940.
- 9.Shannon R D. (1970) Cell dimensions of rare earth orthorhodites". , Acta Crystallographica Section B 26(4), 447-449.
- 10.B A Hunter, C J Howard. (1998) RIETICA. A Computer Program for Rietveld Analysis of X-Ray and Neutron Powder Diffraction Patterns". , Rietica
- 11.J I Langford, Wilson A J C. (1978) Scherrer after sixty years: A survey and some new results in the determination of crystallite size". , Journal of Applied Crystallography 2(11), 102-113.
- 13.A.E lRais, Enas I Baghni and Abdunnaser M Etorki,(2018) "Removal of Methylene Violet from Aqueous Solutions Using BaSr2NbO5.5". , Journal of Analytical & Bioanalytical Techniques 5(8), 338-345.
- 14.Awin A, Mahmoud A El-Rais, Abdunnaser M Etorki, Mokhtar M Abobaker, Mawada S Alzorgani et al. (2021) . Removal of Methyl Violet from Aqueous Solutions using Sr2ANbO5.5(A= Ca+2, Sr+2& Ba+2), International Journal of progressive sciences and technologies 26(1), 67-73.
